A study published in issue 5 of Nature Catalysis showed how a team of scientists from Stanford University created a synthetic catalyst that produces chemicals such as living enzymes. This discovery may result in the industrial-scale transformation of natural gas into low-cost methanol.
Scientific research
For years, scientists have been wondering about the possibility of recreating artificial chemicals and fuels on an industrial scale to match the performance of natural counterparts. In this regard, a team from the SLAC National Accelerator Laboratory have developed a synthetic catalyst of palladium and porous polymers that reproduces enzymes as a living being. The X-ray laboratory experiment showed that the reaction stops when the polymers fill up with CO2, as do the enzymes. By controlling this mechanism, it can be used to produce methanol.
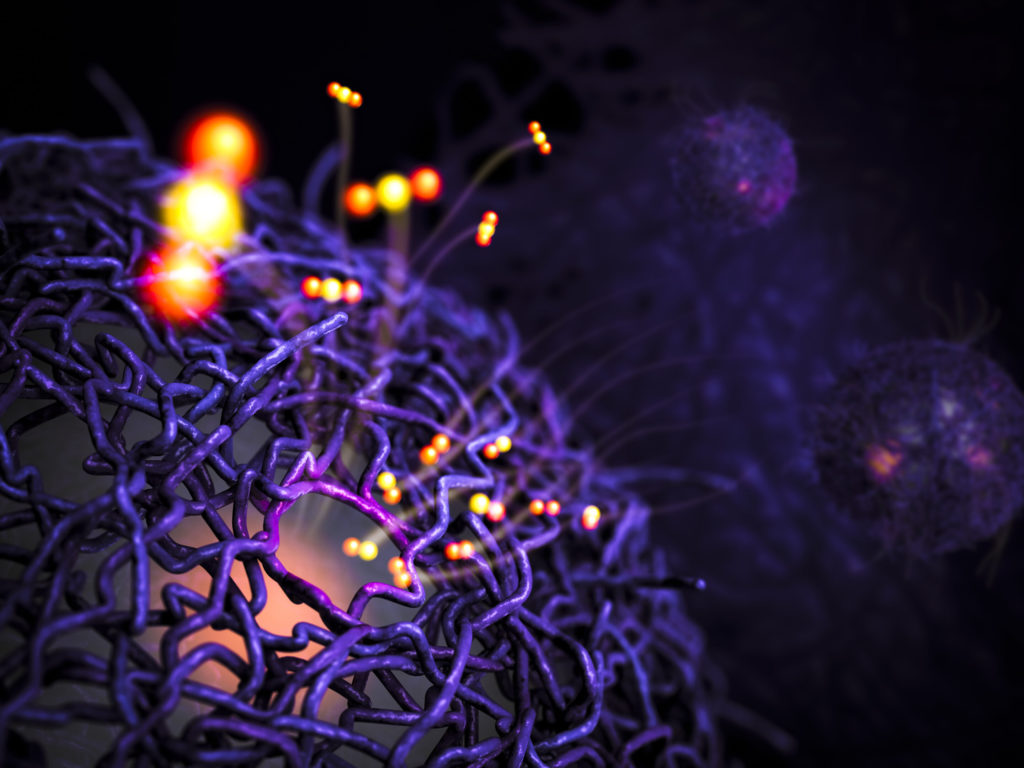
The practical implications
The above mentioned production of methanol comes from a conversion of the most common methane (CH4). Flagged as an economical alternative to petrol, methane is a special remark as it is the basis of the mechanism regulating synthetic catalysts. The future idea is to convert large quantities of methane into low-cost methanol through an artificial catalyst. In this way, there would be a collapse in energy demand and related emissions regulating this mechanism.
The most concrete perspective instead embraces the order of ideas according to which further studies may give rise to the production of artificial enzymes.
If you liked the article, browse our website and like the Facebook page 🙂